


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 17-5-2021
Date: 29-12-2015
Date: 19-12-2015
|
Anhydrides
“Anhydride” means “without water,” and many anhydrides are present in nature. Anhydrides are easily hydrated in the presence of water to acids or bases. Inorganic anhydrides include SO3, P2O5, CaO, Na2O, etc. Acid anhydrides are the most important anhydrides in biochemistry. An acid anhydride [I] is formed by eliminating a molecule of water from two acids (Scheme 1):
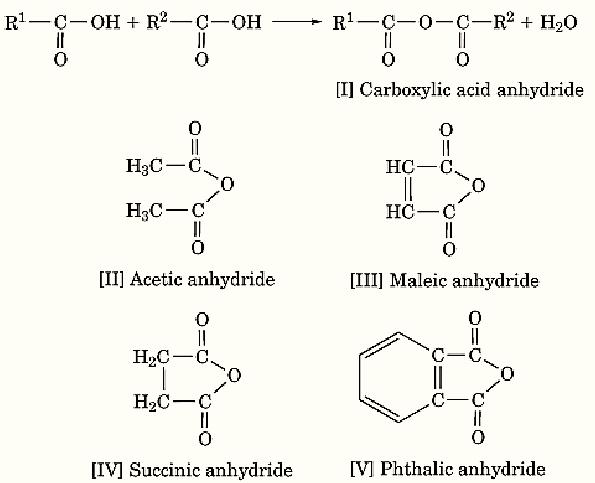
Adding a molecule of water reverses the reaction. Practically, a carboxylic acid anhydride is prepared by the nucleophilic displacement of chloride ion from acyl chlorides by carboxylate ion.
Acid anhydrides are reactive and are good acylating agents. They react with water to yield acids, with amino groups to yield amides, and with alcohols to yield esters. Acid anhydrides are utilized as reactive intermediates in organic syntheses, such as peptide synthesis, and sometimes as enzyme-substrate intermediates. Acid anhydrides are unstable and therefore have large standard free energies of hydrolysis. The two terminal phosphate linkages in ATP are anhydride linkages, and acid anhydrides play important roles in bioenergetics. Acid anhydrides are widely employed for chemical modification of proteins, particularly of amino groups. The acetylation reaction by an acid anhydride with amino groups in a protein proceeds so easily that this reaction is suitable for the radiolabeling of a protein with a radioisotope-labeled acid anhydride (1). Acetic anhydride [II], maleic anhydride [III], and succinic anhydride [IV] are frequently employed in chemical reactions and chemical modifications of proteins. Phthalic anhydride [V] is an aromatic anhydride and is a good leaving group.
Acetic Anhydride
Acetic anhydride is the most important acid anhydride. Its boiling point is 139.6°C, and it has a characteristic penetrating odor. It is widely employed in organic syntheses. It is used to introduce the acetate group into organic compounds and is called an acetylating agent. To acetyle amino groups in a protein (2), prepare a protein solution (2 to 10%, w/v) in half-saturated sodium acetate. Over a period of 1 h at 0°C, add, in five equal portions, a weight of acetic anhydride equal to that of the protein, and continue stirring for an additional hour.
References
1. T. N. M. Schumacher, and T. J. Tsomides (1995) In Current Protocols in Protein Science (J. E. Coligan et al., eds.), Wiley, pp. 3.3.11–3.3.12.
2. T. Imoto and H. Yamada (1989) In Protein Structure: A Practical Approach (T. E. Creighton, ed.), IRL Press, Oxford, UK, pp. 247–277.

|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
سماحة السيد الصافي يؤكد ضرورة تعريف المجتمعات بأهمية مبادئ أهل البيت (عليهم السلام) في إيجاد حلول للمشاكل الاجتماعية
|
|
|