


 الفيزياء الكلاسيكية
الفيزياء الكلاسيكية
 الكهربائية والمغناطيسية
الكهربائية والمغناطيسية
 علم البصريات
علم البصريات
 الفيزياء الحديثة
الفيزياء الحديثة
 النظرية النسبية
النظرية النسبية
 الفيزياء النووية
الفيزياء النووية
 فيزياء الحالة الصلبة
فيزياء الحالة الصلبة
 الليزر
الليزر
 علم الفلك
علم الفلك
 المجموعة الشمسية
المجموعة الشمسية
 الطاقة البديلة
الطاقة البديلة
 الفيزياء والعلوم الأخرى
الفيزياء والعلوم الأخرى
 مواضيع عامة في الفيزياء
مواضيع عامة في الفيزياء|
Read More
Date: 9-3-2016
Date: 23-3-2021
Date: 21-3-2021
|
The Periodic Table
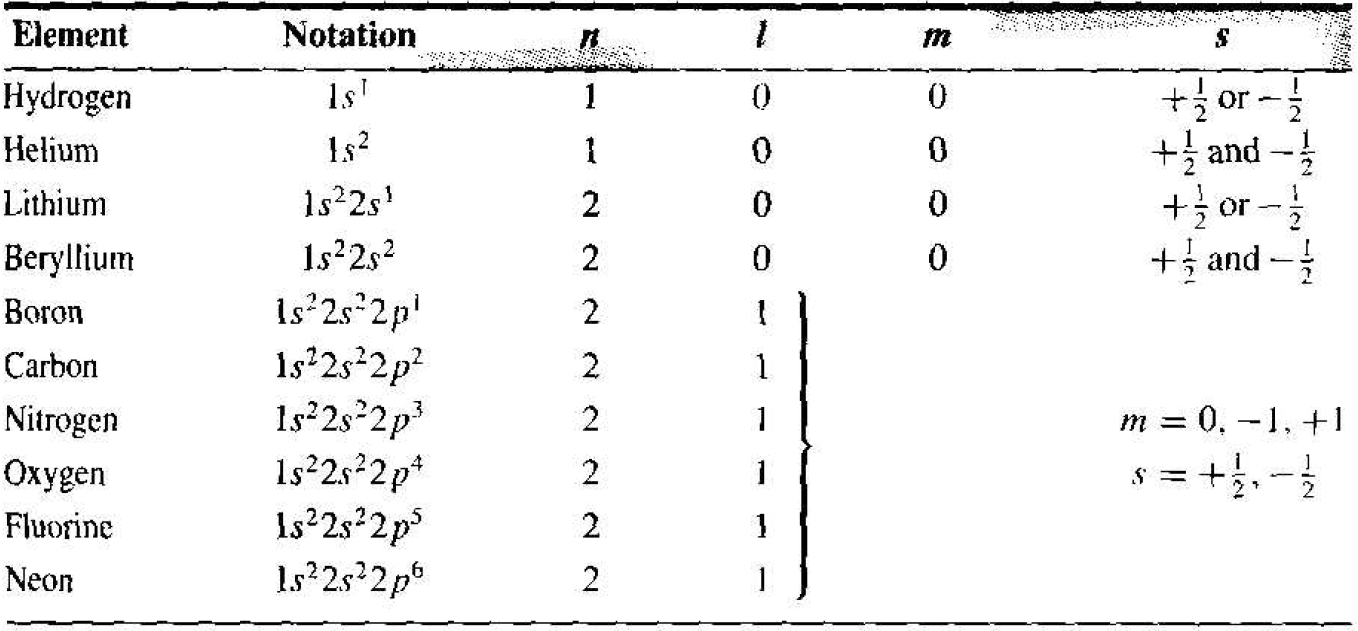
The initial portion of the periodic table of elements may he determined by using the results of the one-electron atom plus two additional concepts. The first concept needed is that of electron spin. The electron has an intrinsic angular momentum, or spin. which is quantized and may take on one of two possible values. The spin is designated by a quantum numbers, which has a value of s = +1/2 or s = - 1/2. We now have four basic quantum numbers: n, l, m, and s.
The second concept needed is the Pauli exclusion principle. The Pauli exclusion principle states that, in any given system (an atom, molecule, or crystal), no two electrons may occupy the same quantum state. In an atom, the exclusion principle means that no two electrons may have the same set of quantum numbers. We will see that the exclusion principle is also an important factor in determining the distribution of electrons among available energy states in a crystal.
Table 1.1 shows the first few elements of the periodic table. For the first element. hydrogen, we have one electron in the lowest energy state corresponding to n = 1. Both quantum numbers l and m must be zero. However, the electron can take one either spin factor +1/2 or -1/2. For helium, two electrons may exist in the lowest energy state. For this case, l = m = 0, so now both electron spin states are occupied and the lowest energy shell is full. The chemical activity of an element is determined primarily by the valence, or outermost, electrons. Since the valence energy shell of helium is full, helium does not react with other elements and is in inert element.
The third element, lithium, has three electrons. The third electron must go into the second energy shell corresponding to n = 2. When n = 2, the quantum number l may be 0 or 1, and when l = 1, the quantum number m may be – 1,0, or +1. In each case, the electron spin factor may be +1/2 or -1/2. For n = 2, then, there are eight possible quantum states. Neon has ten electrons. Two electron, are in the n = 1 energy shell and eight electrons are in the n = 2 energy shell. The second energy shell is now full, which means that neon is also an inert element.
From the solution of Schrodinger's wave equation for the one electron atom, plus the concepts of electron spin and the Pauli exclusion principle, we can begin to build up the periodic table of elements. As the atomic numbers of the elements increase, electrons will begin to interact with each other, so that the buildup of the periodic table will deviate somewhat from the simple method.
Table 1.1 Initial portion of the periodic table



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|