


 الفيزياء الكلاسيكية
الفيزياء الكلاسيكية
 الكهربائية والمغناطيسية
الكهربائية والمغناطيسية
 علم البصريات
علم البصريات
 الفيزياء الحديثة
الفيزياء الحديثة
 النظرية النسبية
النظرية النسبية
 الفيزياء النووية
الفيزياء النووية
 فيزياء الحالة الصلبة
فيزياء الحالة الصلبة
 الليزر
الليزر
 علم الفلك
علم الفلك
 المجموعة الشمسية
المجموعة الشمسية
 الطاقة البديلة
الطاقة البديلة
 الفيزياء والعلوم الأخرى
الفيزياء والعلوم الأخرى
 مواضيع عامة في الفيزياء
مواضيع عامة في الفيزياء|
Read More
Date: 28-3-2021
Date: 9-5-2017
Date: 22-3-2021
|
UNCERTAINTY PRINCIPLE
After this discussion of the wave properties of matter, it is appropriate to approach the most fundamental principle of quantum mechanics, the Uncertainty Principle. According to this principle, no matter how accurately the variables of a quantum mechanical system are determined, a fundamental uncertainty concern their actual magnitude will always remain. It is not possible to gain absolute deterministic information concerning a quantum system. This principle provides an absolute limitation on a determination of the value of a given quantum variable.
For example, consider the double slit experiment. In order to determine which slit the electron passes through, let us place a detector at slit 1 that shines a light or any other radiation on the electron to determine whether the electron passes through the slit (position of electron), as shown in Figure 1.
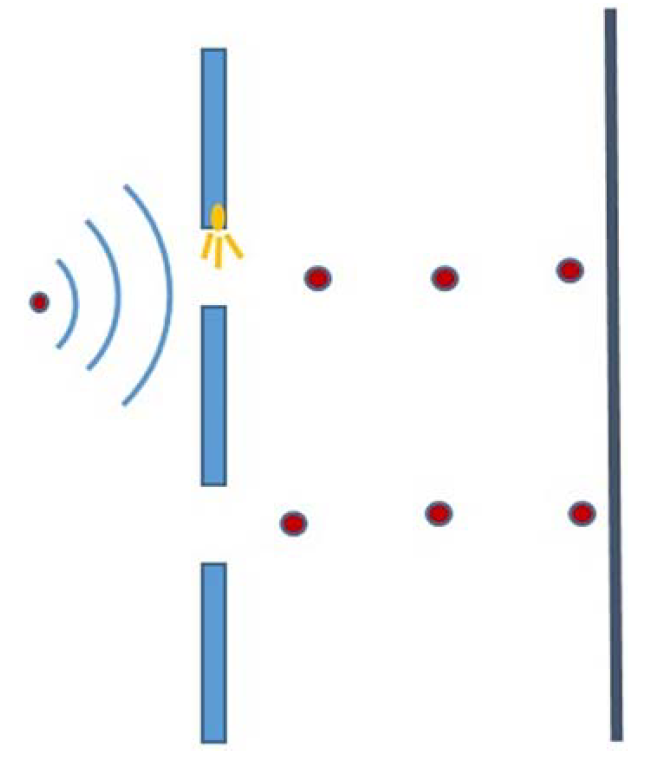
FIGURE 1: The above schematic illustrates that when a detector is placed on one of the slits, the interference pattern vanishes. Instead, two bright spots are observed.
Since light is comprised of photons, as light is shone on the electron that passes through the slit 1, the collision between the photon and the electron knocks the electron from its path. The photon from the light source becomes scattered which can be registered as a detection of an electron. The kick that an electron receives from the photon changes its momentum. This changed momentum cannot be exactly known and is uncertain. However, the position of the electron has become certain.
The scattered photon provides the information about the location of the electron entering through slit 1. It is the fundamental rule of nature if the position of a quantum system has the least uncertainty then momentum has the most uncertainty. Since the momentum and the wavelength of the wave associated with the electron are related by De-Broglie’s relation, the wavelength becomes negligible as momentum becomes uncertain. Hence, all the electrons whose positions are determined follow the straight path because there is no probability wave associated with them and they strike the screen at approximately the same spot. The interference of the electron waves cannot be observed and so instead two intense spots are observed on the screen as depicted in Figure 2. Mathematically, the wavefunction will no longer be a superimposed wavefunction, but rather a single function, ψ1(x), since, we exactly know exact location of the electron.
Why does the certainty in position result in themomentum being uncertain? This is mainly due to a fundamental principle of nature. The principle that mathematically describes such uncertainty, is known as the Uncertainty Principle.
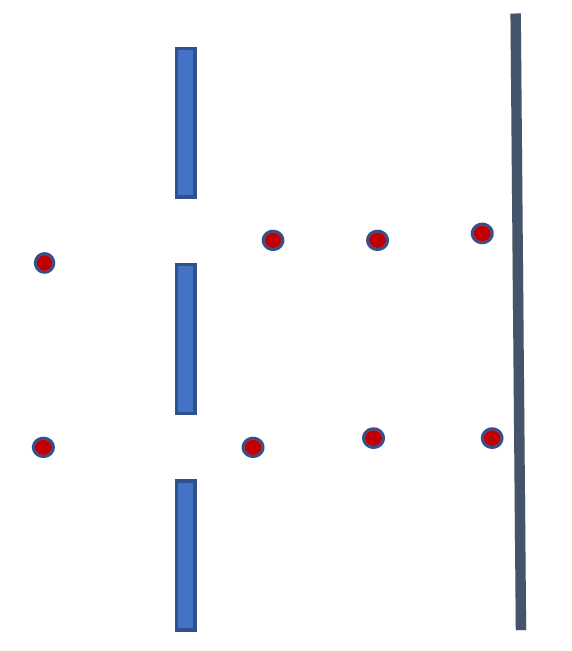
FIGURE 2: The schematic shows that if instead of waves, particles are made to pass through a double-slit then two intense spots will be observed on a screen.



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|