
آخر المواضيع المضافة


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 11-2-2016
Date: 3-1-2017
Date: 29-6-2020
|
Once again, this is not very common for first row transition metal ions. Examples with three different geometries have been identified:
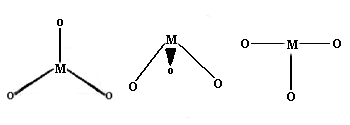
Trigonal planar Geometry: Well known for main group species like CO32- etc., this geometry has the four atoms in a plane with the bond angles between the ligands at 120 degrees.
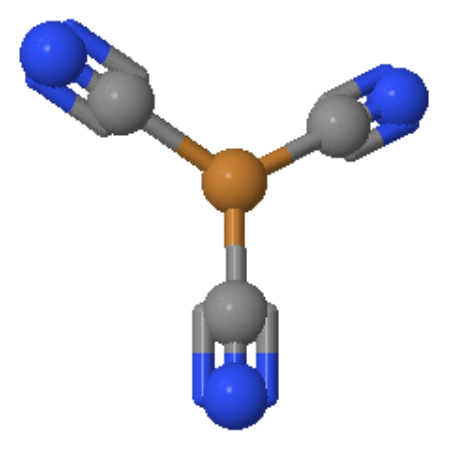
Figure 1: The Trigonal planar [Cu(CN)3]2- system
Trigonal pyramid Geometry: More common with main group ions.
T-shaped: The first example of a rare T-shaped molecule was found in 1977.



|
|
|
|
دراسة: عدم ترتيب الغرفة قد يدل على مشاكل نفسية
|
|
|
|
|
|
|
علماء: تغير المناخ تسبب في ارتفاع الحرارة خلال موسم الحج
|
|
|
|
|
|
|
ضمن مؤتمر الإمام الباقر (عليه السلام).. أكاديمي يناقش التربية العقدية في سلوك الآل المحمدي
|
|
|