


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 5-1-2016
Date: 1-4-2019
Date: 26-12-2016
|
Knowing the States of Matter and Their Changes
Matter is anything that has mass and occupies space. It can exist in one of three classic states: solid, liquid, and gas. When a substance goes from one state of matter to another, the process is called a change of state, or phase change. Some rather interesting things occur during this process, which I explain in this section.
Solids, liquids, and gases
Particles of matter behave differently depending on whether they’re part of a solid, liquid, or gas. As Figure 1 shows, the particles may be organized or clumped, close or spread out. In this section, you look at the solid, liquid, and gaseous states of matter.
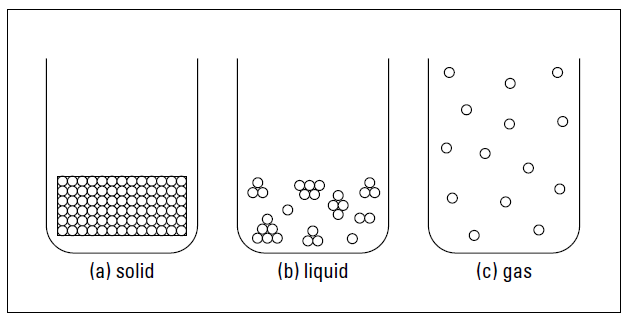
Figure 1: Solid, liquid, and gaseous states of matter.
solid are very close together and aren’t moving around very much (see Figure 1a). That’s because in many solids, the particles are pulled into a rigid, organized structure of repeating patterns called a crystal lattice. The particles in the crystal lattice are still moving but barely — it’s more of a slight vibration. Depending on the particles, this crystal lattice may be of different shapes.
Liquids
Unlike solids, liquids have no definite shape; however, they do have a definite volume, just like solids do. The particles in liquids are much farther apart than the particles in solids, and they’re also moving around much more (see Figure 1b). Even though the particles are farther apart, some particles in liquids may still be near each other, clumped together in
small groups. The attractive forces among the particles aren’t as strong as they are in solids, which is why liquids don’t have a definite shape. However, these attractive forces are strong enough to keep the substance confined in one large mass — a liquid — instead of going all over the place.
Gases
A gas has no definite shape and no definite volume. In a gas, particles are much farther apart than they are in solids or liquids (see Figure 1c), and they’re moving relatively independent of each other. Because of the distance between the particles and the independent motion of each of them, the gas expands to fill the area that contains it (and thus it has no definite shape).



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تستعد لإطلاق الحفل المركزي لتخرج طلبة الجامعات العراقية
|
|
|