


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 7-11-2021
Date: 10-9-2021
Date: 8-10-2021
|
Catecholamines
Dopamine, norepinephrine (NE), and epinephrine (or, adrenaline) are biologically active (biogenic) amines that are collectively termed catecholamines. Dopamine and NE are synthesized in the brain and function as neurotransmitters. Epinephrine is synthesized from NE in the adrenal medulla.
1. Function: Outside the CNS, NE and its methylated derivative, epinephrine, are hormone regulators of carbohydrate and lipid metabolism. NE and epinephrine are released from storage vesicles in the adrenal medulla in response to fright, exercise, cold, and low levels of blood glucose. They increase the degradation of glycogen and triacylglycerol as well as increase blood pressure and the output of the heart. These effects are part of a coordinated response to prepare the individual for stress and are often called the “fight-or-flight” reactions.
2. Synthesis: The catecholamines are synthesized from tyrosine, as shown in Figure 1. Tyrosine is first hydroxylated by tyrosine hydroxylase to form L-3,4-dihydroxyphenylalanine (DOPA) in a reaction analogous to that described for the hydroxylation of phenylalanine . The tetrahydrobiopterin (BH4)-requiring enzyme is abundant in the CNS, the sympathetic ganglia, and the adrenal medulla, and it catalyzes the ratelimiting step of the pathway. DOPA is decarboxylated in a reaction requiring PLP to form dopamine, which is hydroxylated by dopamine β-hydroxylase to yield NE in a reaction that requires ascorbic acid (vitamin C) and copper. Epinephrine is formed from NE by an N-methylation reaction using S-adenosylmethionine (SAM) as the methyl donor .
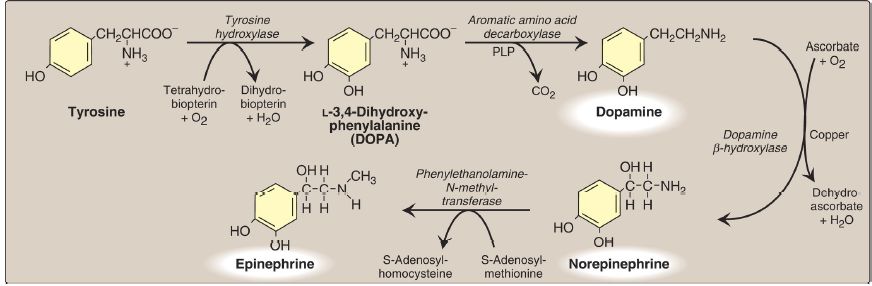
Figure 1: Synthesis of catecholamines. [Note: Catechols have two adjacent hydroxyl groups.] PLP = pyridoxal phosphate.
Parkinson disease, a neurodegenerative movement disorder, is due to insufficient dopamine production as a result of the idiopathic loss of dopamine-producing cells in the brain. Administration of L-DOPA (levodopa) is the most common treatment, because dopamine cannot cross the blood–brain barrier.
3. Degradation: The catecholamines are inactivated by oxidative deamination catalyzed by monoamine oxidase (MAO) and by Omethylation catalyzed by catechol-O-methyltransferase (COMT) using SAM as the methyl donor (Fig. 2). The reactions can occur in either order. The aldehyde products of the MAO reaction are oxidized to the corresponding acids. The products of these reactions are excreted in the urine as vanillylmandelic acid (VMA) from epinephrine and NE and homovanillic acid (HVA) from dopamine. [Note: VMA and the metanephrines are increased with pheochromocytomas, rare tumors of the adrenal gland characterized by excessive production of catecholamines.]
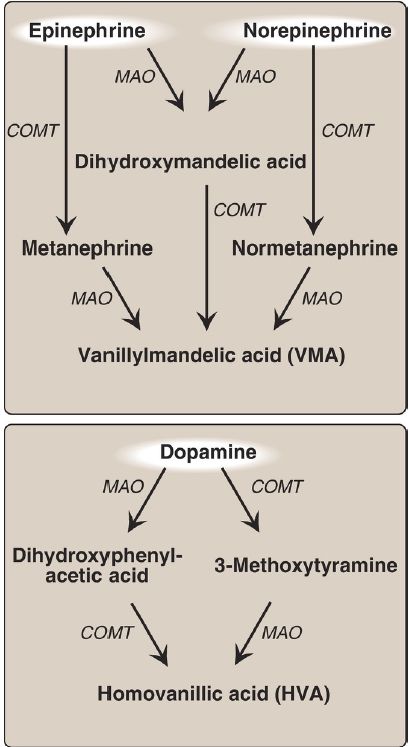
Figure 2: Metabolism of the catecholamines by catechol-O-methyltranferase (COMT) and monoamine oxidase (MAO). [Note: COMT requires Sadenosylmethionine.]
4. Monoamine oxidase inhibitors: MAO is found in neural and other tissues, such as the intestine and liver. In the neuron, this enzyme oxidatively deaminates and inactivates any excess neurotransmitter molecules (NE, dopamine, or serotonin) that may leak out of synaptic vesicles when the neuron is at rest. MAO inhibitors (MAOI) may irreversibly or reversibly inactivate the enzyme, permitting neurotransmitter molecules to escape degradation and, therefore, both to accumulate within the presynaptic neuron and to leak into the synaptic space. This causes activation of NE and serotonin receptors and may be responsible for the antidepressant action of MAOI.

|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تستعد لإطلاق الحفل المركزي لتخرج طلبة الجامعات العراقية
|
|
|