


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 9-9-2020
Date: 11-9-2020
Date: 25-9-2020
|
General Reactions of Carbonyl Compounds
A “nucleophilic addition reaction” (of a carbonyl compound) involves the initial attack of a nucleophile on the slightly positive carbonyl‑carbon atom to form a tetrahedral intermediate.
In a “nucleophilic acyl substitution reaction,” a nucleophile attacks the carbonyl carbon of a carboxylic acid derivative
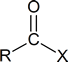
and initially produces a tetrahedral intermediate. The intermediate then reacts by expelling the leaving group, X, thereby forming a new carboxylic acid derivative.
In an “alpha substitution reaction” of a carbonyl compound, one of the hydrogen atoms attached to a carbon atom adjacent to the carbonyl group is removed and replaced by some other group.
Alpha substitution reactions of carbonyl compounds involve the “enol” form of the compound. Keto‑enol tautomerism was briefly introduced in Section 9.4. Review this section if necessary.
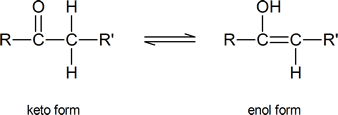
As you can see from the above diagram, the enol form has a hydroxyl group attached to one of the s p2 ‑hybridized carbon atoms of a carbon‑carbon double bond. Removal of the proton from the hydroxyl group produces a resonance‑stabilized “enolate anion”:
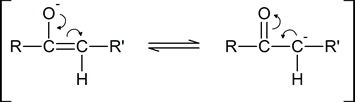
A “carbonyl condensation reaction” is one in which two carbonyl‑ containing molecules condense together (i.e., join together), often with the elimination of water. The reaction involves the formation of an enolate anion from one carbonyl‑containing molecule, followed by the subsequent nucleophilic attack by this enolate anion on the carbonyl carbon atom of the second molecule.



|
|
|
|
5 علامات تحذيرية قد تدل على "مشكل خطير" في الكبد
|
|
|
|
|
|
|
لحماية التراث الوطني.. العتبة العباسية تعلن عن ترميم أكثر من 200 وثيقة خلال عام 2024
|
|
|