


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 7-7-2016
Date: 21-7-2020
Date: 11-3-2017
|
Oxoacids of selenium and tellurium
Selenous acid, H2SeO3, may be crystallized from aqueous solutions of SeO2 and gives rise to two series of salts containing the [HSeO3]- and [SeO3] 2- ions. In aqueous solution, it behaves as a weak acid: pKa(1) ≈ 2.46, pKa(2( ≈ 7:31. Heating salts of [HSeO3]- generates diselenites containing ion 1.1. Tellurous acid, H2TeO3, is not as stable as H2SeO3 and is usually prepared in aqueous solution where it acts as a weak acid: pKa(1) ≈ 2.48, pKa(2) ≈ 7.70. Most tellurite salts contain the [TeO3] 2- ion.
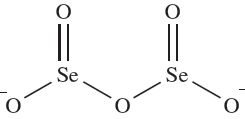

(1.1)
Oxidation of H2SeO3 with 30% aqueous H2O2 yields selenic acid, H2SeO4, which may be crystallized from the solution. In some ways it resembles H2SO4, being fully dissociated in aqueous solution with respect to loss of the first proton. For the second step, pKa =1.92. It is a more powerful oxidant than H2SO4, e.g. it liberates Cl2 from concentrated HCl. Reaction in the solid state between Na2SeO4 and Na2O (2 :1 molar equivalents) leads to Na6Se2O9. This formula is more usefully written as Na12(SeO6)(SeO4)3, showing the presence of the octahedral [SeO6]6- ion which is stabilized in the crystalline lattice by interaction with eight Na ions. The [SeO5]4- ion has been established in Li4SeO5 and Na4SeO5. The formula, H6TeO6 or Te(OH)6, and properties of telluric acid contrast with those of selenic acid. In the solid, octahedral molecules (1.2) are present and in solution, it behaves as a weak acid: pKa)1( = 7.68, pKa)2( = 11.29. Typical salts include those containing [Te)O((OH)5]- and [Te(O)2(OH)4]2- and the presence of the [TeO4]2- ion has been confirmed in the solid state structure of Rb6[TeO5][TeO4].

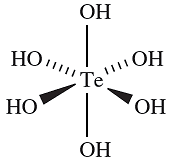
(1.2)



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تستعد لإطلاق الحفل المركزي لتخرج طلبة الجامعات العراقية
|
|
|