
آخر المواضيع المضافة


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 5-7-2020
Date: 5-6-2020
Date: 17-1-2018
|
Estimation of Shielding: Slater Rules
.
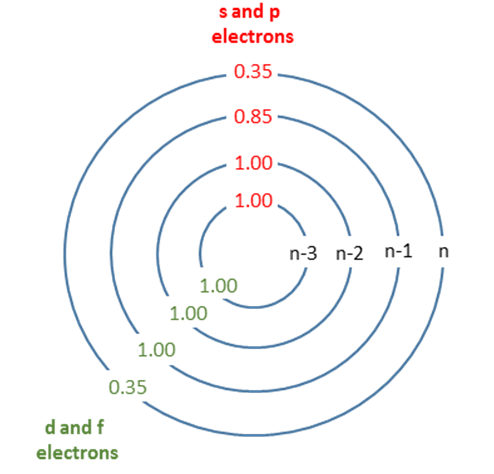
Figure 1 : Graphical depiction of Slaters rules with shielding constants indicated.
Shielding happens when electrons in lower valence shells (or the same valence shell) provide a repulsive force to valence electrons, thereby negating some of the attractive force from the positive nucleus. Ele
ctrons really close to the atom (n-2 or lower) pretty much just look like protons, so they completely negate. As electrons get closer to the electron of interest, some more complex interactions happen that reduce this shielding.
| Group | Other electrons in the same group | Electrons in group(s) with principal quantum number n and azimuthal quantum number < l | Electrons in group(s) with principal quantum number n-1 | Electrons in all group(s) with principal quantum number < n-1 |
|---|---|---|---|---|
| [1s] | 0.30 | - | - | - |
| [ns,np] | 0.35 | - | 0.85 | 1 |
| [nd] or [nf] | 0.35 | 1 | 1 | 1 |
The shielding numbers in Table 1 were derived semi-empirically (i.e., derived from experiments) as opposed to theoretical calculations.



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|