


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 25-7-2018
Date: 25-7-2018
Date: 13-12-2019
|
Natural α-Amino Acids
Hydrolysis of proteins by boiling aqueous acid or base yields an assortment of small molecules identified as α-aminocarboxylic acids. More than twenty such components have been isolated, and the most common of these are listed in the following table. Those amino acids having green colored names are essential diet components, since they are not synthesized by human metabolic processes. The best food source of these nutrients is protein, but it is important to recognize that not all proteins have equal nutritional value. For example, peanuts have a higher weight content of protein than fish or eggs, but the proportion of essential amino acids in peanut protein is only a third of that from the two other sources. For reasons that will become evident when discussing the structures of proteins and peptides, each amino acid is assigned a one or three letter abbreviation.
|
Natural α-Amino Acids |
|
|

The R-substituent in this structure is the remaining structural component that varies from one amino acid to another, and in proline R is a three-carbon chain that joins the nitrogen to the alpha-carbon in a five-membered ring. Applying the Cahn-Ingold-Prelog notation, all these natural chiral amino acids, with the exception of cysteine, have an S-configuration.
For the first seven compounds in the left column the R-substituent is a hydrocarbon. The last three entries in the left column have hydroxyl functional groups, and the first two amino acids in the right column incorporate thiol and sulfide groups respectively. Lysine and arginine have basic amine functions in their side-chains; histidine and tryptophan have less basic nitrogen heterocyclic rings as substituents. Finally, carboxylic acid side-chains are substituents on aspartic and glutamic acid, and the last two compounds in the right column are their corresponding amides.
The formulas for the amino acids written above are simple covalent bond representations based upon previous understanding of mono-functional analogs. The formulas are in fact incorrect. This is evident from a comparison of the physical properties listed in the following table. All four compounds in the table are roughly the same size, and all have moderate to excellent water solubility. The first two are simple carboxylic acids, and the third is an amino alcohol. All three compounds are soluble in organic solvents (e.g. ether) and have relatively low melting points. The carboxylic acids have pKa's near 4.5, and the conjugate acid of the amine has a pKa of 10. The simple amino acid alanine is the last entry. By contrast, it is very high melting (with decomposition), insoluble in organic solvents, and a million times weaker as an acid than ordinary carboxylic acids.
|
|||||||||||||||||||||||||||||||||||||||||||
These differences all point to internal salt formation by a proton transfer from the acidic carboxyl function to the basic amino group. The resulting ammonium carboxylate structure, commonly referred to as a zwitterion, is also supported by the spectroscopic characteristics of alanine.
|
CH3CH(NH2)CO2H |
|
CH3CH(NH3)(+)CO2(–) |
Since amino acids, as well as peptides and proteins, incorporate both acidic and basic functional groups, the predominant molecular species present in an aqueous solution will depend on the pH of the solution. In order to determine the nature of the molecular and ionic species that are present in aqueous solutions at different pH's, we make use of the Henderson - Hasselbalch Equation, written below. Here, the pKa represents the acidity of a specific conjugate acid function (HA). When the pH of the solution equals pKa, the concentrations of HA and A(-) must be equal (log 1 = 0).

The titration curve for alanine, shown below, demonstrates this relationship. At a pH lower than 2, both the carboxylate and amine functions are protonated, so the alanine molecule has a net positive charge. At a pH greater than 10, the amine exists as a neutral base and the carboxyl as its conjugate base, so the alanine molecule has a net negative charge. At intermediate pH's the zwitterion concentration increases, and at a characteristic pH, called the isoelectric point (pI), the negatively and positively charged molecular species are present in equal concentration. This behavior is general for simple (difunctional) amino acids. Starting from a fully protonated state, the pKa's of the acidic functions range from 1.8 to 2.4 for -CO2H, and 8.8 to 9.7 for -NH3(+). The isoelectric points range from 5.5 to 6.2. Titration curves show the neutralization of these acids by added base, and the change in pH during the titration.
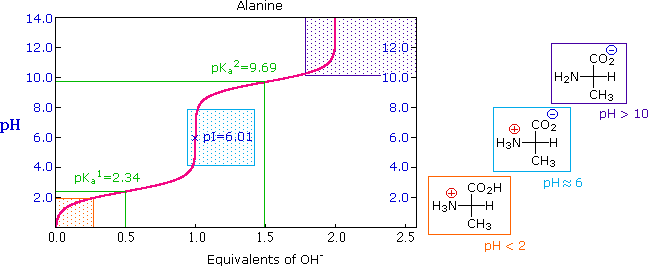
Titration curves for many other amino acids may be examined at a useful site provided by The University of Virginia in Charlottesville.
The distribution of charged species in a sample can be shown experimentally by observing the movement of solute molecules in an electric field, using the technique of electrophoresis. For such experiments an ionic buffer solution is incorporated in a solid matrix layer, composed of paper or a crosslinked gelatin-like substance. A small amount of the amino acid, peptide or protein sample is placed near the center of the matrix strip and an electric potential is applied at the ends of the strip, as shown in the following diagram. The solid structure of the matrix retards the diffusion of the solute molecules, which will remain where they are inserted, unless acted upon by the electrostatic potential. In the example shown here, four different amino acids are examined simultaneously in a pH 6.00 buffered medium. To see the result of this experiment, click on the illustration. Note that the colors in the display are only a convenient reference, since these amino acids are colorless.

|
pKa Values of Polyfunctional Amino Acids |
||||
|
Amino Acid |
α-CO2H |
α-NH3 |
Side Chain |
pI |
|
Arginine |
2.1 |
9.0 |
12.5 |
10.8 |
|
Aspartic Acid |
2.1 |
9.8 |
3.9 |
3.0 |
|
Cysteine |
1.7 |
10.4 |
8.3 |
5.0 |
|
Glutamic Acid |
2.2 |
9.7 |
4.3 |
3.2 |
|
Histidine |
1.8 |
9.2 |
6.0 |
7.6 |
|
Lysine |
2.2 |
9.0 |
10.5 |
9.8 |
|
Tyrosine |
2.2 |
9.1 |
10.1 |
5.7 |
At pH 6.00 alanine and isoleucine exist on average as neutral zwitterionic molecules, and are not influenced by the electric field. Arginine is a basic amino acid. Both base functions exist as "onium" conjugate acids in the pH 6.00 matrix. The solute molecules of arginine therefore carry an excess positive charge, and they move toward the cathode. The two carboxyl functions in aspartic acid are both ionized at pH 6.00, and the negatively charged solute molecules move toward the anode in the electric field. Structures for all these species are shown to the right of the display.
It should be clear that the result of this experiment is critically dependent on the pH of the matrix buffer. If we were to repeat the electrophoresis of these compounds at a pH of 3.80, the aspartic acid would remain at its point of origin, and the other amino acids would move toward the cathode. Ignoring differences in molecular size and shape, the arginine would move twice as fast as the alanine and isoleucine because its solute molecules on average would carry a double positive charge.
As noted earlier, the titration curves of simple amino acids display two inflection points, one due to the strongly acidic carboxyl group (pKa1 = 1.8 to 2.4), and the other for the less acidic ammonium function (pKa2 = 8.8 to 9.7). For the 2º-amino acid proline, pKa2 is 10.6, reflecting the greater basicity of 2º-amines.
Some amino acids have additional acidic or basic functions in their side chains. These compounds are listed in the table on the right. A third pKa, representing the acidity or basicity of the extra function, is listed in the fourth column of the table. The pI's of these amino acids (last column) are often very different from those noted above for the simpler members. As expected, such compounds display three inflection points in their titration curves, illustrated by the titrations of arginine and aspartic acid shown below. For each of these compounds four possible charged species are possible, one of which has no overall charge. Formulas for these species are written to the right of the titration curves, together with the pH at which each is expected to predominate. The very high pH required to remove the last acidic proton from arginine reflects the exceptionally high basicity of the guanidine moiety at the end of the side chain.





|
|
|
|
5 علامات تحذيرية قد تدل على "مشكل خطير" في الكبد
|
|
|
|
|
|
|
لحماية التراث الوطني.. العتبة العباسية تعلن عن ترميم أكثر من 200 وثيقة خلال عام 2024
|
|
|