

علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء التناسقية

الكيمياء الاشعاعية والنووية
SYNTHESIS GAS
المؤلف:
sami matar & Lewis. F. Hatch
المصدر:
Chemistry of PETROCHEMICAL PROCESSES
الجزء والصفحة:
p 122
7-8-2017
2250
SYNTHESIS GAS
Synthesis gas generally refers to a mixture of carbon monoxide and hydrogen. The ratio of hydrogen to carbon monoxide varies according to the type of feed, the method of production, and the end use of the gas. During World War II, the Germans obtained synthesis gas by gasifying coal. The mixture was used for producing a liquid hydrocarbon mixture in the gasoline range using Fischer-Tropsch technology. Although this route was abandoned after the war due to the high production cost of these hydrocarbons, it is currently being used in South Africa, where coal is inexpensive (SASOL, II, and III).
There are different sources for obtaining synthesis gas. It can be produced by steam reforming or partial oxidation of any hydrocarbon ranging from natural gas (methane) to heavy petroleum residues. It can also be obtained by gasifying coal to a medium Btu gas (medium Btu gas consists of variable amounts of CO, CO2, and H2 and is used principally as a fuel gas). Figure 1.1 shows the different sources of synthesis gas.
A major route for producing synthesis gas is the steam reforming of natural gas over a promoted nickel catalyst at about 800°C:
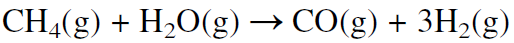
This route is used when natural gas is abundant and inexpensive, as it is in Saudi Arabia and the USA. In Europe, synthesis gas is mainly produced by steam reforming naphtha. Because naphtha is a mixture of hydrocarbons ranging approximately from C5-C10, the steam reforming reaction may be represented using n-heptane:
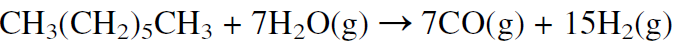
As the molecular weight of the hydrocarbon increases (lower H/C feed ratio), the H2/CO product ratio decreases. The H2/CO product ratio is approximately 3 for methane, 2.5 for ethane, 2.1 for heptane, and less than 2 for heavier hydrocarbons. Noncatalytic partial oxidation of hydrocarbons is also used to produce synthesis gas, but the H2/CO ratio is lower than from steam reforming:

Figure 1.1. The different sources and routes to synthesis gas.
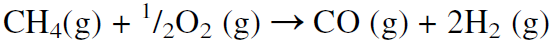
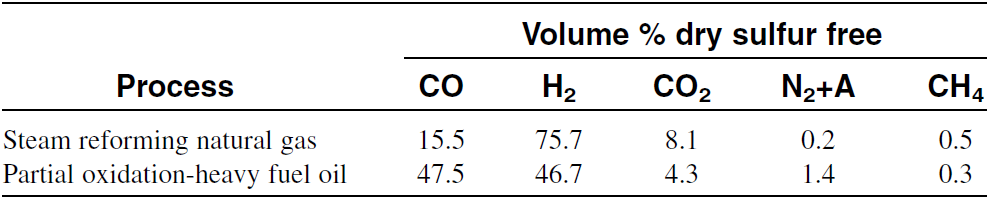
In practice, this ratio is even lower than what is shown by the stoichiometric equation because part of the methane is oxidized to carbon dioxide and water. When resids are partially oxidized by oxygen and steam at 1400–1450°C and 55–60 atmospheres, the gas consists of equal parts of hydrogen and carbon monoxide. Table 1.1 compares products from steam reforming natural gas with products from partial oxidation of heavy fuel oil.
Table 1.1: Composition of synthesis gas from steam reforming natural gas and partial oxidation of fuel oil
 الاكثر قراءة في البترو كيمياويات
الاكثر قراءة في البترو كيمياويات
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















