

علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء التناسقية

الكيمياء الاشعاعية والنووية
Cracking Catalysts
المؤلف:
sami matar & Lewis. F. Hatch
المصدر:
Chemistry of PETROCHEMICAL PROCESSES
الجزء والصفحة:
p 70
24-7-2017
1913
Cracking Catalysts
Acid-treated clays were the first catalysts used in catalytic cracking processes, but have been replaced by synthetic amorphous silica-alumina, which is more active and stable. Incorporating zeolites (crystalline alumina- silica) with the silica/alumina catalyst improves selectivity towards aromatics. These catalysts have both Lewis and Bronsted acid sites that promote carbonium ion formation. An important structural feature of zeolites is the presence of holes in the crystal lattice, which are formed by the silica-alumina tetrahedra. Each tetrahedron is made of four oxygen anions with either an aluminum or a silicon cation in the center. Each oxygen anion with a –2 oxidation state is shared between either two silicon, two aluminum, or an aluminum and a silicon cation.
The four oxygen anions in the tetrahedron are balanced by the +4 oxidation state of the silicon cation, while the four oxygen anions connecting the aluminum cation are not balanced. This results in –1 net charge, which should be balanced. Metal cations such as Na+, Mg2+, or protons (H+) balance the charge of the alumina tetrahedra. A two dimensional representation of an H-zeolite tetrahedra is shown:
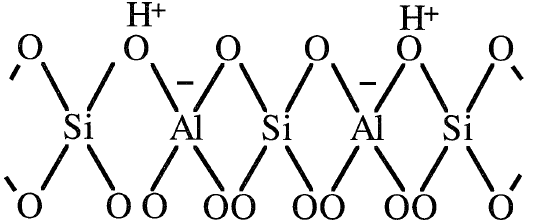
Bronsted acid sites in HY-zeolites mainly originate from protons that neutralize the alumina tetrahedra. When HY-zeolite (X- and Y-zeolites are cracking catalysts ) is heated to temperatures in the range of 400–500°C, Lewis acid sites are formed.
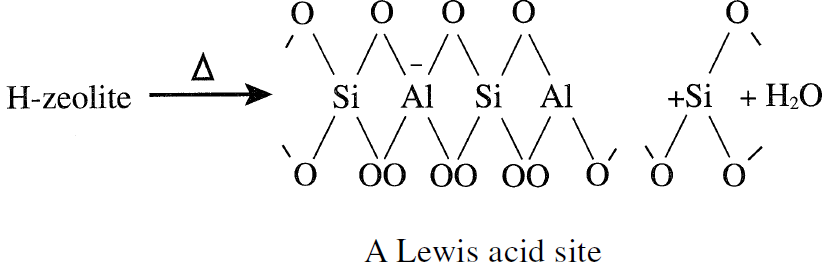
Zeolites as cracking catalysts are characterized by higher activity and better selectivity toward middle distillates than amorphous silica-alumina catalysts. This is attributed to a greater acid sites density and a higher adsorption power for the reactants on the catalyst surface.
The higher selectivity of zeolites is attributed to its smaller pores, which allow diffusion of only smaller molecules through their pores, and to the higher rate of hydrogen transfer reactions. However, the silicaalumina matrix has the ability to crack larger molecules. Hayward and Winkler have recently demonstrated the importance of the interaction of the zeolite with the silica-alumina matrix. In a set of experiments using gas oil and rare earth zeolite/silica-alumina, the yield of gasoline increased when the matrix was used before the zeolite. This was explained by the mechanism of initial matrix cracking of large feedstock
molecules to smaller ones and subsequent zeolite cracking of the smaller molecules to products. Aluminum distribution in zeolites is also important to the catalytic activity. An inbalance in charge between the silicon atoms in the zeolite framework creates active sites, which determine the predominant reactivity and selectivity of FCC catalyst. Selectivity and octane performance are correlated with unit cell size, which in turn can be correlated with the number of aluminum atoms in the zeolite framework.
Deactivation of zeolite catalysts occurs due to coke formation and to poisoning by heavy metals. In general, there are two types of catalyst deactivation that occur in a FCC system, reversible and irreversible. Reversible deactivation occurs due to coke deposition. This is reversed by burning coke in the regenerator. Irreversible deactivation results as a combination of four separate but interrelated mechanisms: zeolite dealumination, zeolite decomposition, matrix surface collapse, and contamination by metals such as vanadium and sodium.
Pretreating the feedstocks with hydrogen is not always effective in reducing heavy metals, and it is expensive. Other means that proved successful are modifying the composition and the microporous structure of the catalyst or adding metals like Sb, Bi or Sn, or Sb-Sn combination. Antimony organics have been shown to reduce by 50% gas formation due to metal contaminants, especially nickel.
 الاكثر قراءة في البترو كيمياويات
الاكثر قراءة في البترو كيمياويات
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















