


 الفيزياء الكلاسيكية
الفيزياء الكلاسيكية
 الكهربائية والمغناطيسية
الكهربائية والمغناطيسية
 علم البصريات
علم البصريات
 الفيزياء الحديثة
الفيزياء الحديثة
 النظرية النسبية
النظرية النسبية
 الفيزياء النووية
الفيزياء النووية
 فيزياء الحالة الصلبة
فيزياء الحالة الصلبة
 الليزر
الليزر
 علم الفلك
علم الفلك
 المجموعة الشمسية
المجموعة الشمسية
 الطاقة البديلة
الطاقة البديلة
 الفيزياء والعلوم الأخرى
الفيزياء والعلوم الأخرى
 مواضيع عامة في الفيزياء
مواضيع عامة في الفيزياء|
Read More
Date: 7-11-2020
Date: 29-3-2017
Date: 28-3-2017
|
Radioactive Equilibrium
Radioactive equilibrium exists when a radioactive nuclide is decaying at the same rate at which it is being produced. Since the production rate and decay rate are equal, the number of atoms present remains constant over time.
An example of radioactive equilibrium is the concentration of sodium-24 in the coolant circulating through a sodium-cooled nuclear reactor. Assume that the sodium-24 is being produced at a rate of 1 x 10 atoms 6 per second. If the sodium-24 were stable and did not decay, the amount of sodium-24 present after some period of time could be calculated by multiplying the production rate by the amount of time. Plotting the amount of material present would result in the graph in Figure 1.
However, sodium-24 is not stable, and it decays with a half-life of 14.96 hours. If no sodium-24 is present initially and production starts at a rate of 1 x 106 atoms per second, the rate of decay will initially be zero because there is no sodium-24 present to decay. The rate of decay of sodium-24 will increase as the amount of sodium-24 increases.
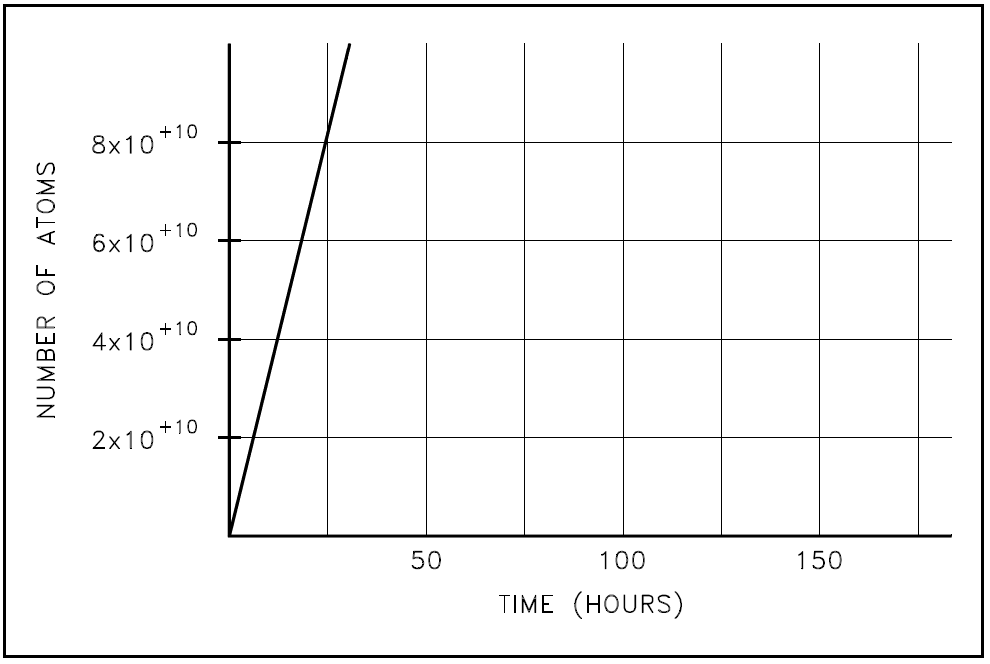
Figure 1: Cumulative Production of Sodium-24 Over Time
The amount of sodium-24 present will initially increase rapidly, then it will increase at a continually decreasing rate until the rate of decay is equal to the rate of production. It is possible to calculate how much sodium-24 will be present at equilibrium by setting the production rate (R) equal to the decay rate (λ N).

where:
R = production rate (atoms/second)
λ = decay constant (second-1)
N = number of atoms
It is possible to calculate the equilibrium value for sodium-24 being produced at a rate of 1 x 106 atoms/second.

The development of the equation to calculate how the amount of sodium-24 changes over time as it approaches the equilibrium value is beyond the scope of this handbook. However, the equation is presented below.
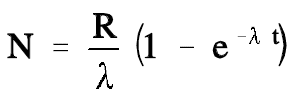
This equation can be used to calculate the values of the amount of sodium-24 present at different times. As the time increases, the exponential term approaches zero, and the number of atoms present will approach R/λ. A plot of the approach of sodium-24 to equilibrium is shown in Figure 2.

Figure 2: Approach of Sodium-24 to Equilibrium



|
|
|
|
4 أسباب تجعلك تضيف الزنجبيل إلى طعامك.. تعرف عليها
|
|
|
|
|
|
|
أكبر محطة للطاقة الكهرومائية في بريطانيا تستعد للانطلاق
|
|
|
|
|
|
|
العتبة العباسية المقدسة تبحث مع العتبة الحسينية المقدسة التنسيق المشترك لإقامة حفل تخرج طلبة الجامعات
|
|
|