
Atomic Radiation
 المؤلف:
Roger J Blin-Stoyle, FRS
المؤلف:
Roger J Blin-Stoyle, FRS
 المصدر:
Physics of Particles, Matter and the Universe
المصدر:
Physics of Particles, Matter and the Universe
 الجزء والصفحة:
P 81
الجزء والصفحة:
P 81
 22-5-2016
22-5-2016
 2765
2765
Atomic Radiation
We have seen that radiation is emitted when an electron jumps from one energy level to another having lower energy. It may have been knocked into the higher level in a discharge tube by

Figure 1.1: (a) Spontaneous emission, (b) absorption and (c) stimulated emission of atomic radiation with hv = EA –EB.
energetic electrons moving across the tube. The emission of radiation is illustrated in figure 1.1(a) for a transition between two levels labelled A and B having energies E, and E,. The associated frequency I, is related to the energy difference between the levels by the usual formula given in the diagram. This is referred to as spontaneous emission. For small energy differences the frequencies lie in the optical range. However sometimes the energy difference can be large. This happens, for example, in an x-ray tube when atoms are bombarded by high-energy electrons. These can knock out electrons from filled energy levels deep down in the atom leaving, as it were, a vacancy or hole which the exclusion principle allows to be filled. An electron from a much higher energy level can jump into the hole and the resultant radiation can then have a high frequency in the x-ray range. If an electron is in an energy level (e.g. A in figure 1.1) and has the possibility of making a transition to a lower level, the point (in time) at which it will make the transition cannot be predicted. All that quantum mechanics can tell us is that on average it will spend a certain time (referred to as its mean life) in that level. Typically, in an atomic transition. such lifetimes are of the order of 10-8s. This is an uncertainty in the time at which an event happens and there is another important associated uncertainty relation of the form
(time uncertainty) × (energy uncertainty) 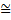 h
h
which means that the emitted photons in a transition do not have the exact frequency given by the formula because of the (small) uncertainty in the energy of the level A relating to its lifetime. The lines in an atomic spectrum therefore have a small width in their frequency. It is by measuring this width and using the above uncertainty relation that the lifetime of atomic states can be estimated. Another possible radiation process is absorption (see figure 1.1(b)) in which a photon of the right frequency excites an electron from the level B to the level A. This means that: if light with a spread of frequencies passes through a gas the gas atoms will absorb light of certain frequencies when electrons are excited to higher levels. In turn this leads to dark lines in the spectrum of the light that has passed through the gas. This is called an absorption spectrum and is clearly characteristic of the gas. Absorption spectroscopy is an important analytical process and has been used, for example, to identify atoms (and molecules) in space through which sunlight and starlight has passed. Stimulated emission of radiation can also occur when an electron in an atom is in an excited level, for example level A in figure 1.1(c), and radiation falls on it. If this radiation has the frequency appropriate for a transition to the level B, then it stimulates the electron to make that transition and so the end product is two photons. The important thing about this process is that the two waves associated with the photons are in phase (coherent) in that their crests and troughs coincide and they also travel in the same direction. This means that they reinforce each other very strongly. Of course, if the state A has a typical atomic lifetime then it will, in any case, make this transition spontaneously and stimulated emission will be hard to observe. However some special states (referred to as metastable) can have a very long lifetime (as long as s) and there are ways of ‘pumping’ many atoms into such states. Passing light of the appropriate frequency through such a ‘pumped up’ collection of atoms and reflecting it backwards and forwards then results in a great deal of stimulated emission and an immensely powerful resultant coherent light wave is produced. This is the principle underlying the operation of a laser where the word derives from the phrase Light Amplification by Stimulated Emission of Radiation.
 الاكثر قراءة في الفيزياء الذرية
الاكثر قراءة في الفيزياء الذرية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة
