


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 3-12-2015
Date: 6-5-2021
Date: 3-3-2021
|
Complementation
The term complementation usually refers to the situation where two defective genomes in the same cell together support a normal or nearly normal phenotype, each supplying the function that the other lacks. Another meaning of the term, in the context of DNA manipulation, is the repair of a mutational deficiency in an organism or cell culture by artificially introducing a gene that supplies the missing function. This is a potent way of cloning genes selected according to function. In either sense, complementation provides a way of discriminating between gene functions and of defining genes as functional units.
1.Complementation and the definition of the functional gene
Classical genetics was based upon clear-cut heritable variants, either induced by mutagenic treatments or, in earlier days, just turning up as “sports” in wild or cultivated populations. The geneticists' favorite organisms, notably maize (Zea mays) and the fruit-fly (Drosophila melanogaster), were diploid. Most mutations were recessive to wild type, which is what would be expected if they mostly represented losses of function that could be more or less adequately supplied if one gene out of two was normally active.
As mutants accumulated, they were attributed to the same or different genes on the basis of two criteria. First, it was supposed that mutations in the same gene were inseparable by recombination. A diploid that carries two different mutant alleles (ie, alternative forms) of a particular gene should never produce nonmutant or double-mutant germ cells by free reassortment or crossing-over at meiosis. According to the second criterion, mutations in the same gene should affect the organism in similar or at least related ways. Similarity or overlap of functional effects was judged by individual appearance (phenotype) and also and more rigorously by failure of the mutant chromosomes to complement each other's defects in the hybrid diploid. (In fact, the term complementation did not come into use until the late 1950s, and I use the word with hindsight).
A classical example of using the complementation criterion, and one of the first in which it was thrown into doubt, involves the sex-linked white (w) gene of Drosophila, one of several genes governing eye color. A considerable number of recessive mutant w alleles had been identified. They diluted the normal red-brown eye pigment to various degrees when the same allele was present on both X-chromosomes (homozygous) in females, or on the single X in males. Females that had two different mutant w alleles (heterozygous) had dilute eye colors generally intermediate between the colors of the two homozygotes. Thus females of constitution white/ apricot (w/wa) have pale apricot eyes, not wild-type red as would be expected if each mutant X-chromosome could provide the pigment-forming function lacking in the other.
In 1952, E. B. Lewis (1) found that, at very low frequency (about 1 in 10,000), w/wa females produce eggs of recombinant types, with neither mutation or both. By the attached-X technique, which need not be described here, he showed that these recombinants arise by crossing-over between the X-chromosomes, just as if white and apricot were mutations at distinct, though very closely linked loci. Accordingly he gave them different symbols, w and a, and rewrote the constitution of the female parent wa+/w+a, where the + superscripts denote the respective wild-type alleles. The remaining anomaly was what Lewis called the cis /trans position effect (Fig. 1). Two kinds of doubly heterozygous constitution could be compared, the original so-called trans arrangement wa+/w+a, that has the two mutations on opposite chromosomes, and the cis arrangement, w+ a+/wa. They had identical overall gene content, but the former had pale apricot eyes and the latter was fully wild type. w+ and a+ could act cooperatively to promote normal levels of eye pigment only when they were together on the same chromosome. Lewis called their relationship pseudoallelic: allelic according to their failure to complement each other but nonallelic according to their ability to recombine. A formally identical situation at the Drosophila lozenge locus had already been reported by M. M. Green (2).
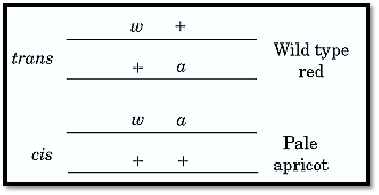
Figure 1. Lewis's (1) cis-trans comparison using two mutations in the Drosophila melanogaster X-linked white (w( gene. The mutations white (w) and apricot (wa, shown here as a) are each recessive to wild type in females but do not complement one another in trans. By this criterion they are allelic in spite of being separable at low frequency by recombination.
For Lewis and Green at that time, the recombination criterion for allelism took priority, and the reason for the position effect was a matter for speculation. Before long, however, high-resolution recombination analysis of series of mutants in the microbial world, first in the fungus Aspergillus nidulans (3) and then in the bacterial virus (bacteriophage) T4 (4), revealed that pseudoallelism, in Lewis's sense, was much more the rule than the exception. It was rather rare for independently occurring mutations to be “truly” allelic. Almost all pairwise combinations of noncomplementing mutants showed some low frequency of recombination when crossed together. Soon afterward, the same was shown to be true in two bacterial species, Escherichia coli and Salmonella typhimurium, (5) which have no regular diploidy or meiosis but other means exist for obtaining partial diploids and tests for complementation and recombination. In one example after another, it was shown that most mutations within functional genes are at different mutually recombinable sites which, by different methods in different organisms, could be mapped in a closely spaced linear sequence that was continuous with the much longer sequence of the whole chromosome.
Following his very extensive analysis of the rII series of mutants in phage T4 (4), S. Benzer proposed replacing the word gene with a new term, cistron, meaning the unit of function as defined by Lewis's cis/trans comparison. When the cis and trans phenotypes are identical (wild type, if the mutants involved were recessive), the mutations are in different cistrons, affecting different functions. If trans is mutant and cis normal (or relatively so) the mutants are in the same cistron. The concept has been universally accepted, but the term cistron has dropped out of use. Now “gene” is generally used in the same sense, and the full cis-trans test is rarely applied. The theoretical reason for the comparing trans (m1 + / + m2) with cis (m1m2/ (+ + , rather than just trans with the wild type, is that it controls the possibility that the cumulative effect of two heterozygous mutations in different genes could be a sub-wild phenotype, even though each is individually recessive to its wild-type allele. In practice, however, recessive alleles usually remain recessive even when several of them are present together, and so the trans/wild comparison, the simple complementation test, is usually deemed sufficient.
Although diploidy provides the means for complementation testing in higher plants and animals and also in budding yeast, alternative methods, described under Complementation tests, have to be used for habitually haploid organisms, such as bacteria, viruses, and most fungi.
1.1. Complementation and gene cloning in yeasts
Cloning wild-type alleles of yeast genes with metabolic functions and mutating to give auxotrophic phenotypes, became straightforward following the development of the Saccharomyces two-micron(-2µm) plasmid as a cloning vehicle (7). This plasmid also replicates in the other major experimental yeast species, the fission yeast Schizosaccharomyces pombe. It has been most useful as a component of a series of hybrid shuttle vectors, capable of replication either in yeast or in Escherichia coli. These usually incorporate the replicative origin of the E. coli plasmid ColE1 and a gene for antibiotic resistance (e.g., to tetracycline) that can be selected for in the bacterial host.
To select and clone a yeast gene, fragments of total yeast DNA, produced by digestion with a restriction endonuclease (or sometimes by sonication), are ligated into the closed-loop, double-stranded DNA of a shuttle vector and introduced en masse by one of the standard transformation procedures into mutant yeast cells, which then are plated on a medium on which they grow to form colonies only if their mutational deficiency has be repaired. The first yeast genes to be cloned were selected because they conferred on auxotrophic (nutritionally exacting) mutants the ability to form colonies on a minimal (unsupplemented) growth medium. But any gene that mutates to give a conditional no-growth phenotype can be cloned in essentially the same way. Temperature sensitive mutants have been a particularly rich source of cloned genes. Almost any essential protein can be mutated to a temperature-sensitive form, functional at, say, 30°C but not at 36°C. Mutant cells grown at the permissive temperature are transformed with a gene “library” and selected for repair of function at the restrictive temperature.
The shuttle vector bearing the selected gene replicates autonomously either in yeast or after transfer to E. coli. The bacterial host is more efficient for mass propagation of the clone. When sufficiently amplified, the cloned gene is cut out of the vector, and its structure and function are analyzed in various ways, most obviously by DNA sequencing. The general method is outlined in Fig. 2.
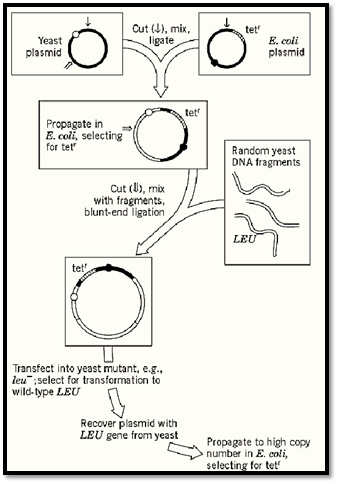
Figure 2. Cloning and amplifying a yeast gene in a shuttle vector selected by its complementation of a yeast auxotrophic (leucine-requiring) mutant. Open and filled circles indicate replication origins for propagation in yeast and E. coli, respectively. Based on Beggs' first demonstration of the method (7).
The complementation of yeast mutants is used for cloning yeast genes and also genes from such distant organisms as Drosophila and humans. One good example is cloning the human equivalent of the Schizosaccharomyces pombe (fission yeast) CDC2 gene (8). S. pombe temperature-sensitive cdc2 mutants cannot complete their cell cycle at elevated temperature (36°C) because the mutant cdc2 gene product, a protein kinase that provides a signal essential for initiating cell division, is inactivated at this temperature. Transformation of mutant cells with a human cDNA library cloned in a replicating plasmid led to the isolation of a few colonies that harbor hybrid plasmids carrying the desired cDNA sequence. The cloning plasmid used in this instance differs from that illustrated in Fig. 2 in that its replication in fission yeast depends on a DNA sequence from the monkey SV40 virus, which replicates well in S. pombe, not on the yeast 2-mm plasmid. To clone mammalian gene sequences in yeasts, it is generally necessary to use cDNA rather than genomic DNA. Mammalian introns are not effectively spliced out in yeast cells and, in any case, often make the gene too long for cloning in one piece.
1.2. In Neurospora
Although yeast species that have a single-cell, colony-forming habit and are accessible to replicating plasmids are especially convenient for cloning genes, the complementation principle can also be applied to filamentous fungi and has been particularly successful in Neurospora crassa. This species has the disadvantage that it has no convenient plasmid that stably replicates without integration into the chromosomes. Consequently, although auxotrophic mutants are “rescued” by transformation with fragments of total Neurospora DNA, it is not so easy to recover the transforming sequences from the transformed cultures. This difficulty was overcome by sib selection (9.(
The method is making a library of Neurospora DNA sequences in some suitable vector (sufficient in number so that it is probable that the great majority of genes are represented), dividing the clones into a number of pools, and testing DNA isolated from each pool for its ability to repair the mutant of interest. Then a pool that works (and there is usually at least one) is subdivided into smaller pools for further tests, and then to still smaller pools, until the hunt is narrowed down to a small number of clones that can be tested individually. Though obviously laborious, the method has been very successful. It has been used for cloning genes of metabolic function and also for the loci (actually gene complexes) that govern fungal mating type by complementation of mating-deficient mutants (10, 11) .
1.3. In Drosophila
Many gene functions involved with basic cellular processes are common to yeast and mammals. Then it is not surprising to find that others, more to do with tissue differentiation, are common to most animals. Because the genetic control of development is better understood in Drosophila than in any other animal, it is of interest to ask whether some of the relatively well-characterized Drosophila genes have mammalian equivalents. Flies cannot be used for the kind of mass screening of gene libraries that is possible with yeast, but it is not difficult to find out whether some particular mouse gene or cDNA clone complements a Drosophila mutant.
It is relatively easy to get exotic DNA into the Drosophila genome by inserting it into the transposable P element and injecting the construct into early embryos. Thus the mouse gene M33, cloned on the basis of its sequence similarity to the Drosophila polycomb (Pc) gene, was successfully introduced into Pc mutant flies (12). The mutants, which usually have sex combs on all legs of the male instead of just on the front pair as in the wild type, were partially normalized by inserting one copy of M33 into their genome and were restored almost to wild type by two copies. Pc protein is one of a group of chromosomal proteins that act together to prevent certain developmentally important genes from being expressed in the wrong places or at the wrong times. This experiment strongly indicates that there is a mammalian gene that has a similar function.
References
1. E. B. Lewis (1952) Proc. Natl. Acad. Sci. USA 3, 953–961.
2. M. M. Green and K. C. Green (1949) Proc. Natl. Acad. Sci. USA 35, 586–591.
3. R. H. Pritchard (1953) Heredity 9, 343–371.
4. S. Benzer (1955) Proc. Natl. Acad. Sci. USA 41, 344–354.
5. P. E. Hartman, J. C. Loper, and D. Serman (1960) J. Gen. Microbiol. 22, 323–368.
6. S. Benzer (1958) In The Chemical Basis of Heredity (W. D. McElroy and B. Glass, eds.), Johns Hopkins Press, Baltimore, pp. 70–93.
7. J. D. Beggs (1978) Nature 275, 104–109.
8. M. G. Lee and P. M. Nurse (1987) Nature 327, 31–33.
9. S. J. Vollmer and C. Yanofsky (1986) Proc. Natl. Acad. Sci. USA 83, 4867–4873.
10. N. L. Glass, J. Grotelueschen, and R. L. Metz (1990) Proc. Natl. Acad. Sci. USA 87, 4912–4916.
11. C. Staben and C. Yanofsky (1990) Proc. Natl. Acad. Sci. USA 87, 4917–4921.
12. J. Muller, S. Gaunt, and P. A. Lawrence (1995) Development 121, 2847–2852.

|
|
|
|
مخاطر خفية لمكون شائع في مشروبات الطاقة والمكملات الغذائية
|
|
|
|
|
|
|
"آبل" تشغّل نظامها الجديد للذكاء الاصطناعي على أجهزتها
|
|
|
|
|
|
|
تستخدم لأول مرة... مستشفى الإمام زين العابدين (ع) التابع للعتبة الحسينية يعتمد تقنيات حديثة في تثبيت الكسور المعقدة
|
|
|